“I had been told that after two years of being cancer-free, I had beaten the disease,” recalls Don Pender, 67, an autoworker from Spring Hill, Tennessee. He thought his struggle against non-Hodgkin lymphoma was in the rearview mirror.
Then, in early 2015, almost five years after showing no signs of the disease, he got some bad news.
“I was shaving, and I felt this lump in my neck. It was quite large. I went back to my doctor, and she checked it out and did a biopsy,” he says. “Then she told me, ‘I’m sorry to tell you this because it usually doesn’t come back.’”
But it had. Pender was told that his options at the regional center had been exhausted, and he was referred to Vanderbilt-Ingram Cancer Center in Nashville for a stem cell transplant using his own cells.
Lymphoma occurs when white blood cells, usually B cells, grow out of control. Although chemotherapy is effective for some people with aggressive non-Hodgkin lymphoma, the cancer often relapses and a stem cell transplant may be the next step. Cancerous immune cells are killed off with radiation or strong chemotherapy and replaced either with a patient’s own previously collected blood-forming stem cells or with those from a matched donor.
Pender underwent the grueling procedure that spring, but within a year, that treatment had also failed.
“I was told that the only thing that I could try was a transplant from my sister or brother, but I had already been told that the donor stuff doesn’t really work all that well,” he says. He was also told that he might have only six months left to live.
As Pender was leaving the office to consider his grim options, the doctor called out, “Wait a second, Don, come back in. Would you be willing to do a trial?”
In March 2016, Pender was offered a chance to join a trial of chimeric antigen receptor T-cell therapy, better known as CAR-T, a customized treatment that uses gene therapy to reprogram a patient’s immune cells to fight cancer. T cells are collected from the blood and shipped to a manufacturing facility, where they are modified, multiplied and sent back to be reinfused into the patient. But in addition to killing cancer cells, the supercharged T cells can also harm healthy cells, leading to potentially life-threatening side effects.
Lymphoma patient Don Pender and Olalekan Oluwole, MBBS, MPHCourtesy of Vanderbilt-Ingram Cancer Center
Jamie Galucci, a 29-year-old paralegal from Stuart, Florida, was first diagnosed in January 2015 with primary mediastinal large B-cell lymphoma, a rare type of lymphoma in which abnormal B cells form a mass behind the breastbone. She received standard rituximab-based chemotherapy at a local hospital, but the treatment failed to completely eradicate the cancer.
Galucci did some research and sought a second opinion from Jason Westin, MD, head of the B-cell lymphoma research team at the University of Texas MD Anderson Cancer Center in Houston.
“After confirmation of cancer activity via a biopsy and a PET scan, my mass began to aggressively grow, and the plan was to get me into remission so I could ultimately undergo a stem cell transplant,” Galucci recalls. “I went home to Florida and underwent two rounds of chemotherapy.”
After returning to Texas for further testing, Galucci also received disappointing news: Her cancer was not responding to the treatment.
“We learned that my mediastinal mass was refractory to the chemotherapy regimen and had grown to approximately 7 centimeters,” she says. “Due to time constraints and not being able to undergo the stem cell transplant, I prepared for a concurrent radiation and chemotherapy regimen. There was a six-day waiting period before I would begin, so I decided to travel home to save money and spend time with family.”
“On my way back to Houston for treatment, I received a call that would forever change my life: The FDA [Food and Drug Administration] had approved the Kite CAR-T immunotherapy trial, and I was moved to the top of the list,” she recalls. “I was told I was a perfect candidate for the treatment. I was very nervous, to say the least, knowing that the side effects could ultimately result in my death, but I had no choice.”
Today—almost two years later—both Pender and Galucci are feeling fine and their cancer is in remission. Pender is enjoying retirement. Galucci is back at work. They are among the first patients treated with Yescarta, the newly approved CAR-T therapy developed by Kite Pharma in Santa Monica, California, now part of Gilead Sciences.

Yescarta trial participant Jamie GalucciEd Susman
On October 18, 2017, the FDA approved Yescarta (also known by the tongue-twisting generic name axicabtagene ciloleucel) for adults with relapsed or refractory large B-cell lymphoma who have unsuccessfully undergone two or more types of treatment. The first CAR-T therapy, Kymriah (also known as tisagenlecleucel), from Novartis, was approved on August 31, 2017, for children and young adults with acute lymphoblastic leukemia.
With the current standard of care, people with aggressive B-cell lymphoma often have an expected survival of around six months. Now these patients have another option.
To understand the bewildering scientific aspects of the CAR-T therapy trial, Pender assembled his own expert team—his son and daughter-in-law, both trained as nurses. They accompanied him to meetings with the researchers to help him understand the process and its potential risks and benefits.
“But in my mind, I didn’t really see any other option that was good for me, so I had pretty much made my decision that I was going to do it,” he recalls.
After he agreed to be in the clinical trial, known as ZUMA-1, Pender had to undergo a battery of tests within a short two-week window of opportunity to determine whether he met the criteria for inclusion. “As luck would have it, the Good Lord was watching over me, and I qualified,” he says.
At Vanderbilt, Pender came under the care of Madan Jagasia, MBBS, MS, coleader of the Translational Research and Interventional Oncology Research Program, and Olalekan Oluwole, MBBS, MPH, the assistant professor of medicine who led the Yescarta trial at the center.
“The first day they put those cells back in me at Vanderbilt, I felt fine. I thought, Hey, this is no big deal,” says Pender. And then he went into a coma for four days and spent 10 days in intensive care.

Sattva Neelapu, MDCourtesy MD Anderson Cancer Center
While it has the potential to be lifesaving for people with limited options, CAR-T therapy is not a stroll in the park, as Pender and Galucci can attest. They both developed cytokine release syndrome—also known as a “cytokine storm”—in which the ramped-up T cells unleash a flood of immune system chemicals, leading to severe side effects, such as high fever, low blood pressure and brain swelling.
“As part of the national team testing this new CAR-T therapy, we have developed specific protocols to manage these patients,” Jagasia says. “Vanderbilt has a group of highly experienced physicians who work closely with subspecialty care teams, and we have immediate access to the intensive care unit.”
Galucci was reinfused with her modified CAR-T cells on November 30, 2015. Her treating doctor, Sattva Neelapu, MD, deputy chair of the Department of Lymphoma and Myeloma at MD Anderson Cancer Center, also had to manage a rough bout of cytokine release syndrome.
“I suffered from cytokine release syndrome, neurotoxicity and seizures, ultimately being forced to learn how to walk, talk and eat again,” Galucci recalls. “But if I had to do it all over again, I would, because, on December 29, 2015, I was informed that I was in remission, and I have stayed in remission since that date. I have also bounced back without any major residual side effects or issues.”
In her case, the comatose condition lasted for two days—the average time for the complication to run its course, according to Neelapu. Kite recommends that patients undergoing treatment with Yescarta be monitored closely for seven days after the infusion. If they live some distance from the hospital, as was the case for both Pender and Galucci, they should prepare to stay nearby for at least four weeks in case life-threatening infections occur.

Madan Jagasia, MBBS, MSCourtesy of Vanderbilt-Ingram Cancer Center
“The immune system is conditioned to fight viruses and bacteria, and when someone has a severe infection, it can cause fevers,” Oluwole explains. “A similar pattern occurs when the modified T cells encounter lymphoma or leukemia cells. There is an immune response that can cause fevers and other side effects.
“The more we use this technology, the better we get at detecting subtle signs earlier, and we can be more proactive in providing interventions to minimize the toxicities. The key is recognizing them early and intervening before the patient gets too sick,” Oluwole continues. “I think it is safe to say these new CAR-T therapies will become part of what we do on a day-to-day basis to treat lymphoma. Because of the way this new immunotherapy works, this technology has actually moved the field further toward eradicating cancer.”
After working through the harrowing experience of the cytokine storm, patients come back for a radiological assessment about four weeks later; these evaluations occur regularly for around two years.
“I felt that the treatment had worked almost immediately,” Galucci recalls. Her feeling was backed up by the first of her tests a month after the initial infusion: “There was no more cancer.”
In Pender’s case, the first scan at 30 days showed that his cancer was reduced by at least half. At three months, the imaging tests showed just a few small spots of residual disease. By six months, his scans were clear—and they have remained that way.
“I knew something good was happening because you can feel your lymph nodes in your neck, and I couldn’t feel anything,” Pender says. After the scan was done, one of the technicians told him, “We are in awe of what we are seeing.”
“When I saw Dr. O, he said the cancer was at least 50 percent gone after just one month. He said the treatment was working and it would keep working for at least six months to a year,” Pender adds. “After six months, it was all gone. I was in remission. I just had another test in September, and it is still all gone.”
“I am Catholic, and I do practice my faith. A lot of my friends prayed for me, so I just believe a lot of that worked,” Pender says. “The people who developed this thing have given me a blessing. I feel good and I have my energy back, although it took a while to get there. I think this will save a lot of lives. I wouldn’t be here today if I hadn’t been in this trial.”
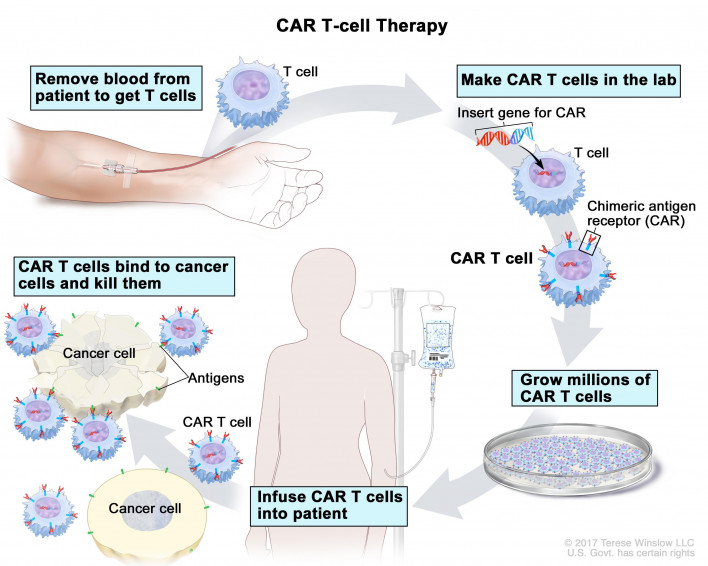
How CAR-T Works
Chimeric antigen receptor T-cell (CAR-T) therapy reprograms T cells to recognize and kill cancer cells. First, a sample of normal T cells is collected using a process called leukapheresis. Blood is removed from a vein in the arm, the T cells are separated out and the rest of the blood is returned to the body.
The T cells are frozen and sent to a manufacturing facility, where they are genetically engineered to create a customized “living drug” for each patient. Using an inactivated virus, lab scientists insert artificial receptors that bind to a specific type of cancer cell. The first two approved CAR-T therapies—Kite’s Yescarta and Novartis’s Kymriah—are designed to recognize the CD19 protein on B cells that grow out of control in lymphoma and leukemia. Researchers are now working on similar therapies that home in on other targets.
The engineered T cells are multiplied in an incubator and shipped back to the local hospital. Before receiving the modified cells, the patient undergoes strong chemotherapy to kill off existing immune cells and make room for the new ones. The whole process takes around two to three weeks.
Studies have shown that a single infusion of reprogrammed T cells can sometimes lead to complete remission that lasts for years. In the ZUMA-1 trial of Yescarta, 72 percent of lymphoma patients had at least a partial response, including 51 percent who achieved complete remission. The first child treated with Kymriah for leukemia remains in remission five years later. But researchers don’t yet know how to predict who will respond, and so far this type of treatment works better for blood cancers than for solid tumors.
Courtesy of National Cancer Institute







1 Comment
1 Comment